30 New Independent Tests Underway: Including Endotoxins (LPS), Heavy Metals & Microbiological Analysis

We currently have 30 new independent laboratory tests underway, including endotoxin (LPS) testing, heavy metal screening, and full microbiological analysis. Every batch is independently verified by our long-term analytical partner Liquilabs s.r.o., using professional-grade analytical methods such as HPLC and LC-MS.
Endotoxin (LPS) testing is the single most important safety requirement when purchasing any peptide. It is not optional, and it should never be hidden under general microbiological claims.

Why This Matters When Buying Peptides
In the research peptide industry, purity alone is not enough. Even peptides that appear clean can still contain dangerous bacterial endotoxins (LPS) — residues from Gram-negative bacteria that are not detected by standard microbial growth tests.

Without proper testing, peptides may contain:
• Endotoxins (LPS) — bacterial residues, even when no live bacteria are present
• Heavy metals
• Microbiological contamination
• Residual solvents
• Incorrect peptide content or concentration
This is why LPS testing must always be listed separately, and never lumped together with general microbiological screening.
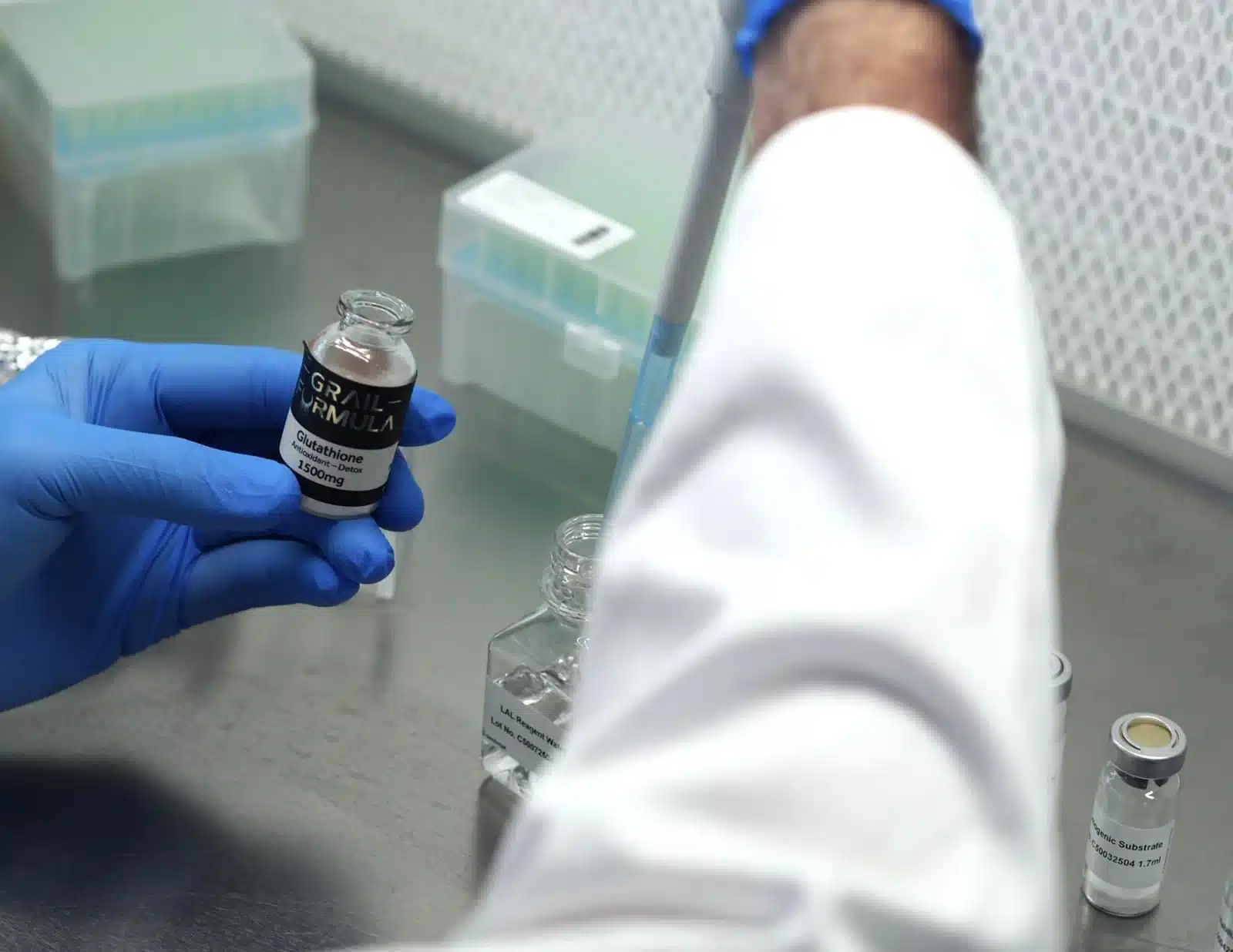
Tested Where It Matters
The images above show our peptides being analyzed at Liquilabs — a specialized European laboratory focused on analytical verification and safety testing. This is where our Certificates of Analysis originate, and where every new batch is validated before release, including endotoxin limits.

Our Standard
• Independent third-party testing
• Dedicated endotoxin (LPS) testing
• Heavy metal screening
• Microbiological analysis
• Ongoing batch verification
• Full analytical transparency

If a peptide supplier cannot clearly show where, how, and which specific safety tests are performed — especially endotoxin testing — that should be considered a red flag.
Verified testing is not optional. Endotoxin testing is the baseline.
